|
6/12/2023 0 Comments Calculate pi of amino acid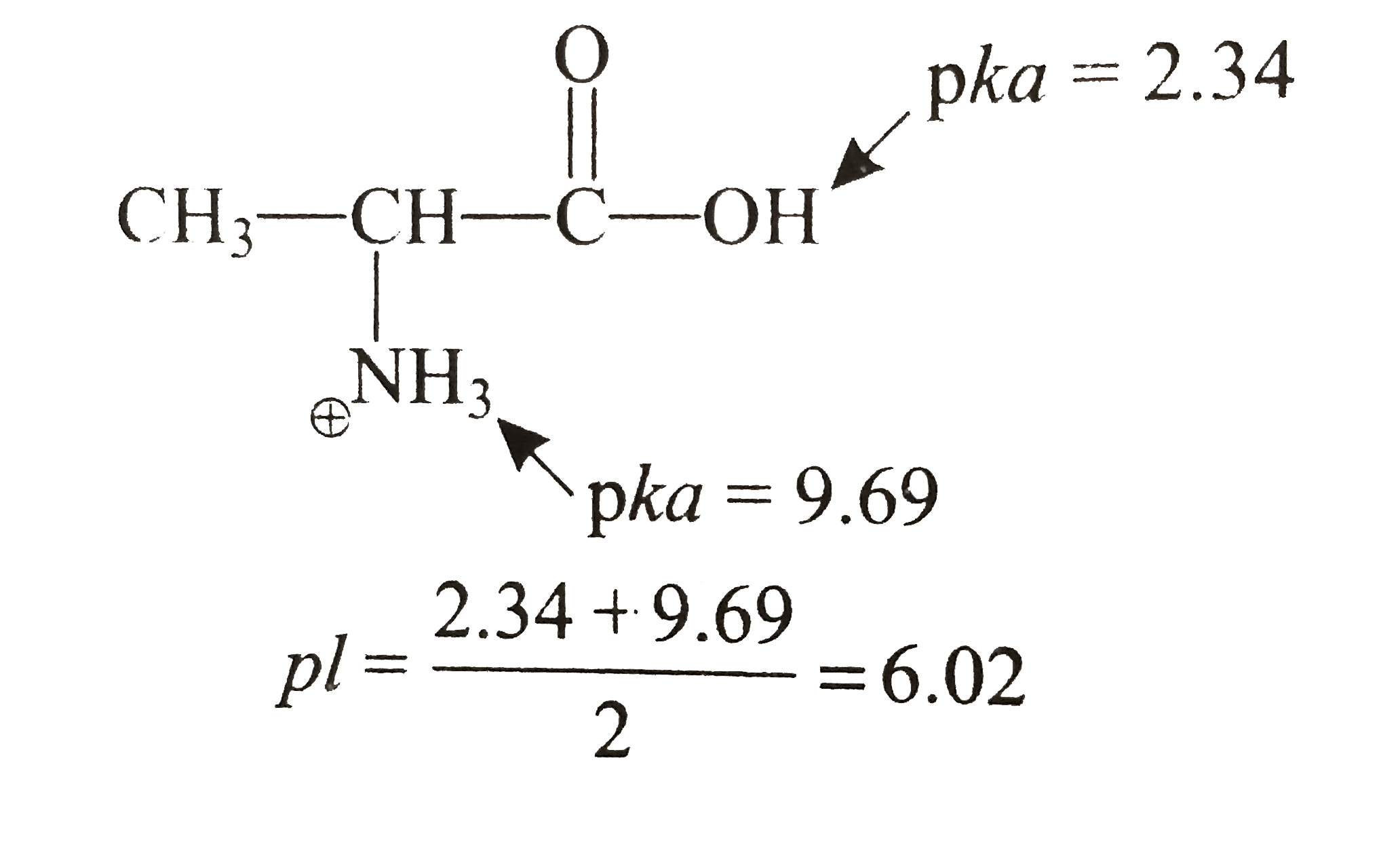
Once all pK values are known, the IEP can be calculated using the following equation: The pK value is a measure of the acidity or basicity of a functional group it is the negative logarithm of the equilibrium constant for dissociation of that group. To calculate the IEP of a molecule or surface, one must first determine the pK values of all functional groups present. The IEP is an important parameter in many fields including biochemistry, protein engineering, and water treatment. The isoelectric point (IEP) is the pH at which a particular molecule or surface carries no net electrical charge. 
Once you know which side will be more charged, you can use that information to figure out what range of pH values will cause that side to be charged.įor example, let’s look at glycine (Gly), whose structure is shown below: In most cases, this will be the side with more oxygens. This can be done by looking at which side has more hydrogen atoms attached to electronegative atoms like oxygen or nitrogen. To calculate the pI, first determine which side of the molecule will be more charged at neutral pH (7). When the pH of a solution equals the pI of an amino acid, the two groups cancel each other out and the amino acid has no net charge. These groups have opposite charges the carboxyl group is acidic and the amine group is basic. The amino acid has two functional groups, the carboxyl group (-COOH) and the amine group (-NH2). The pI of an amino acid can be calculated using its structure. The pI (isoelectric point) is the pH at which a particular amino acid carries no net electrical charge. A pH less than 7 is acidic and greater than 7 is basic. It is measured on a scale from 0 to 14, with 7 being neutral. PH is a measure of the acidity or basicity of an aqueous solution. Conclusion How is Pi of Amino Acid Calculated?.Isoelectric Point of Amino Acids is Used for.How to Calculate Isoelectric Point With 3 Pkas.How to Calculate Isoelectric Point of a Peptide.How To Calculate The Isoelectric Point of Amino Acids and Zwitterions.It also becomes more complicated for polyprotic groups, but all the groups in amino acids are monoprotic with water as a solvent. if one group becomes negatively charged, it becomes more "difficult" for the neighboring group to become negatively charged). This is only an approximation because there might be some cross talk between ionizable groups (i.e. The Henderson-Hasselbalch relationship describing each ionizable group is: I am asking for the expected value of the net charge (which would not be an integer) this number is relevant, for example, for the migration speed of the amino acid (or a protein) in gel electrophoresis or the strength of interaction with ion exchange chromatography media.įor example, a carboxylic acid/carboxylate group at a pH equal to its pKa would have an average charge of minus one half because half of the functional groups would be protonated (charge of zero) and half would be deprotonated (charge of minus one). Thus, when determining the average net charge across the ensemble (or a time-averaged charge of a single particle), you have to take this into account. When the pKa of one group (or more) is close enough to the pH, a fraction of the amino acids will be deprotonated at that group and the other fraction of amino acids will be protonated at that group in solution. When determining the charge of an amino acid, you have to take into consideration the pH and the pKa's of each of these groups. How do I determine the net charge of that amino acid when there are mixed protonation states of one or more of the groups at that pH (pKa of side chain, for example, is really close to the pH)?Īmino acids have terminal carboxyl and amino groups some amino acids have ionizable side chains. I am given an amino acid with an ionizable side chain at a certain pH.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
